Drug Diffusion in the Brain
Modeling Carmustine Diffusion into the Brain as a Function of Time & Location
Understanding the Blood Brain Barrier
This project modeled how Gliadel wafer placement influences carmustine diffusion in the brain after glioblastoma resection. Using analytical diffusion equations and 3D ANSYS simulations, my team quantified how drug concentration and flux vary with distance from the implant and proximity to the blood–brain barrier, highlighting tradeoffs between treatment coverage and healthy‑tissue exposure.
My work centered on background research and ANSYS modeling. I translated clinical and transport‑phenomena literature into model assumptions and boundary conditions, then built finite‑element diffusion simulations showing reduced efficacy with distance from the wafer and drug accumulation near the blood–brain barrier, consistent with theoretical predictions.
The Setup
After glioblastoma resection, recurrence is nearly universal and systemic toxicity limits conventional chemotherapy. Therefore, localized delivery must navigate the brain’s complex anatomy and diffusion constraints to avoid uneven dosing or damage to healthy tissue.
To establish realistic assumptions and clinically relevant boundary conditions for the model, I researched glioblastoma pathology, clinical use of Gliadel, and diffusion in biological media.
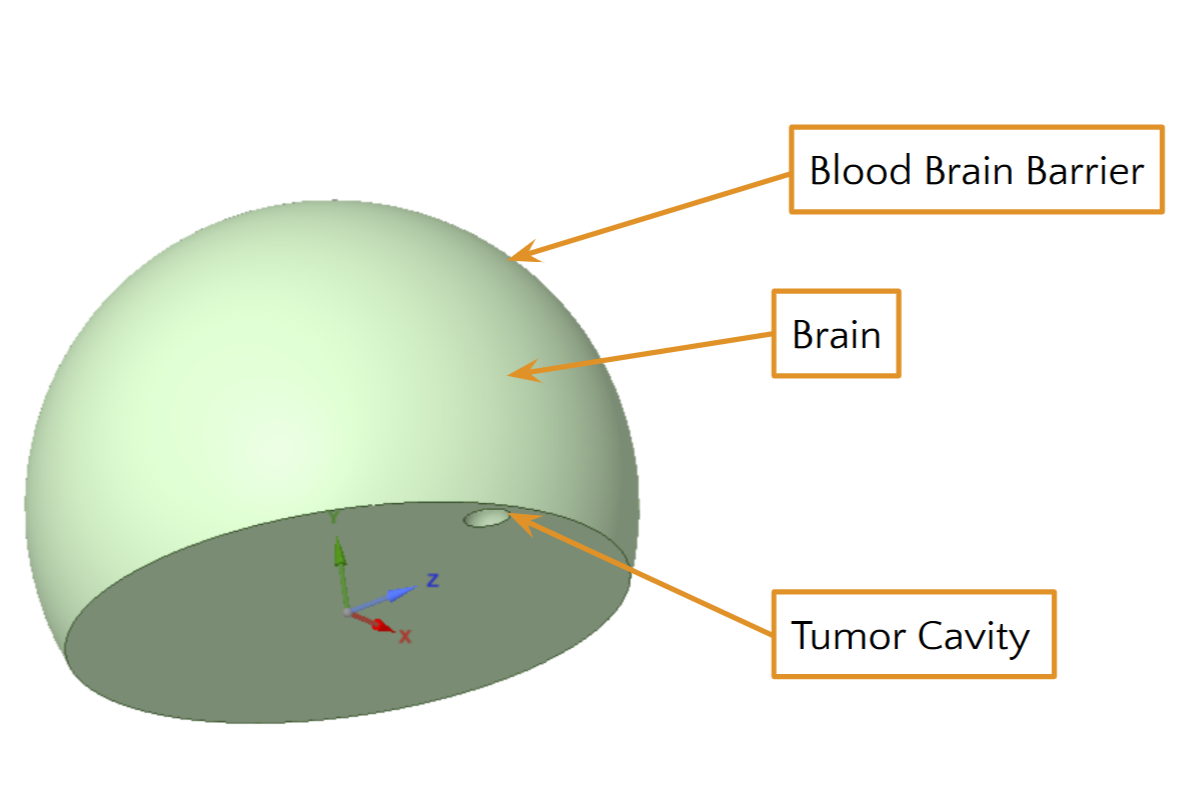
In this project, the brain is defined as a half sphere, with the tumor being placed at some point along the edge.
I developed analytical and ANSYS-based diffusion models using Fick’s Law in spherical coordinates to study how tumor cavity location affects carmustine transport. By comparing ideal infinite-boundary cases with anatomically constrained geometries (treating the cavity as a localized source and the blood–brain barrier as a limiting boundary), I isolated how boundary proximity distorts concentration gradients over time.
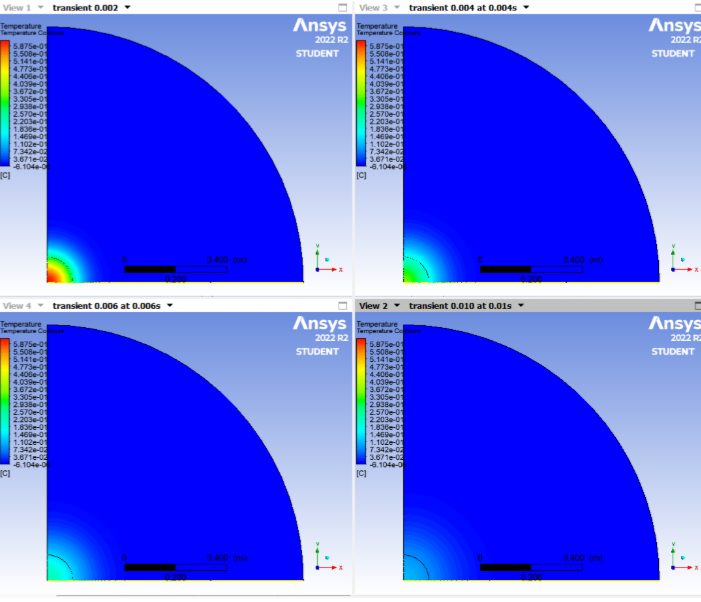
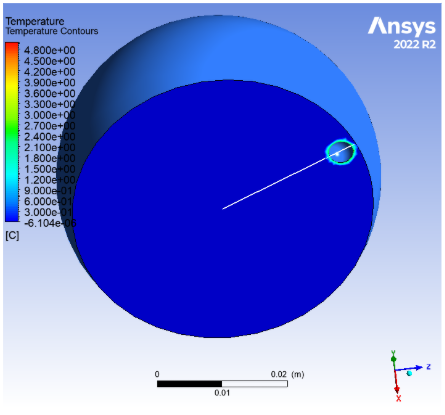
I evaluated the 4-part Ideal model and the systemic real model side by side.
Results
The models showed that carmustine concentration decreases rapidly with distance from the wafer, reducing therapeutic exposure farther from the implantation site. When the tumor cavity was positioned near the blood–brain barrier, diffusion across the boundary was constrained, leading to localized drug accumulation near the interface.
This accumulation highlights a clinically important risk: while localized delivery improves targeting of residual tumor cells, boundary effects can elevate toxicity in adjacent healthy tissue. Overall, the results emphasize that implant location significantly influences both efficacy and safety, underscoring the need for placement-aware planning and improved drug delivery strategies.
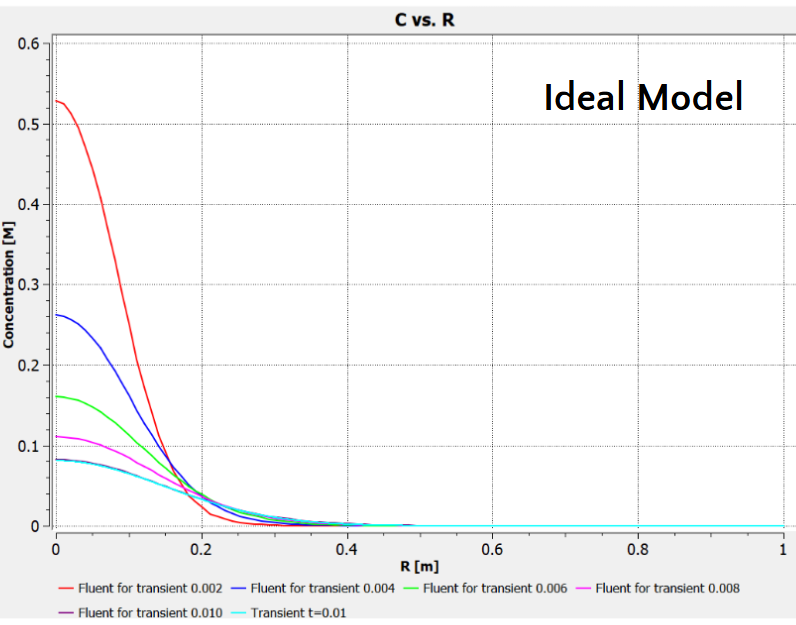
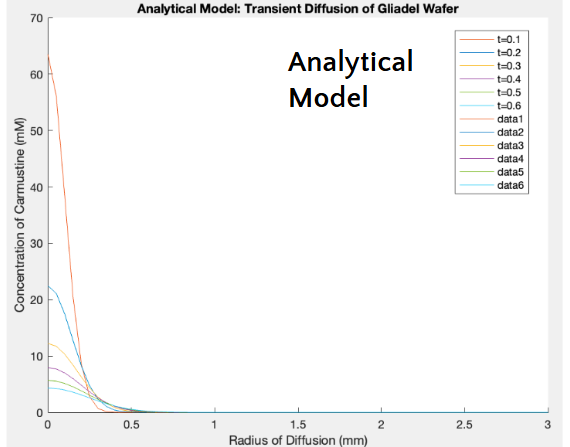
Both the ideal and analytical models are depicted, and they both highlight the same trends - that the concentration of carmustine decreases as the distance from the center of the tumor (R) increases and as time increases.